Most articles on insulin resistance read like a pamphlet: a few vague lines about “cells not responding,” a reminder to “eat healthy,” and not much else. If you’ve ever left a reputable site feeling like you still don’t actually understand what’s happening in your body, you’re not imagining it. Insulin resistance is one of those topics where the public explanation is often technically true but practically useless.
This article is different. We’re going to break down insulin resistance in a way that actually makes it click: what it is at the cellular level, why it develops, why it can hide for years behind “normal” blood sugar, and what the most reliable signs and measurements look like in the real world. We’ll also cover the part that gets missed in almost every oversimplified overview—insulin resistance is not insulin “going bad,” and blaming insulin doesn’t explain the problem. Insulin is doing its job. The issue is that muscle and liver stop responding as well to insulin’s signal, so the pancreas has to turn the volume up just to keep your bloodstream stable.
The big idea you’ll see repeatedly is this: insulin resistance isn’t primarily a “bad food” story. It’s an energy story. When fuel comes in more reliably than it goes out—especially for long stretches—your body has to store more, and storage eventually spills into places it shouldn’t (like liver and muscle). That spillover jams the signal, insulin has to run higher, and the system becomes harder to regulate. Once you understand that, the solutions stop feeling random and start feeling obvious.
By the end of this, you’ll know exactly what insulin resistance is (and what it isn’t), the major drivers that cause it, the symptoms and lab markers that actually matter, and the highest-ROI ways to improve insulin sensitivity—whether you’re just noticing early signs or you’ve been told you’re prediabetic. No fluff, no moralizing, and no vague “eat better” advice—just the mechanisms and the levers that actually move the needle.
What Is Insulin Resistance
Insulin resistance is when your body needs more insulin than it should to keep blood sugar and fuel under control.
The problem is not actually with insulin itself, but with how well your tissues respond to it. After you eat, your pancreas releases insulin as a signal that tells your muscles and liver to pull fuel out of the bloodstream and handle it safely—use it, store it, and keep blood sugar stable.
Over time, those tissues can become less responsive to that signal, often because the system has been dealing with more incoming energy than it’s using over long periods, often chronically. When the message isn’t being heard as clearly, the pancreas compensates by turning up the volume, producing more insulin to get the same effect.
That’s why insulin resistance often begins with normal-looking blood sugar: glucose can stay controlled for years precisely because insulin is working overtime in the background. The real shift is that stability now requires a higher insulin output than it should.
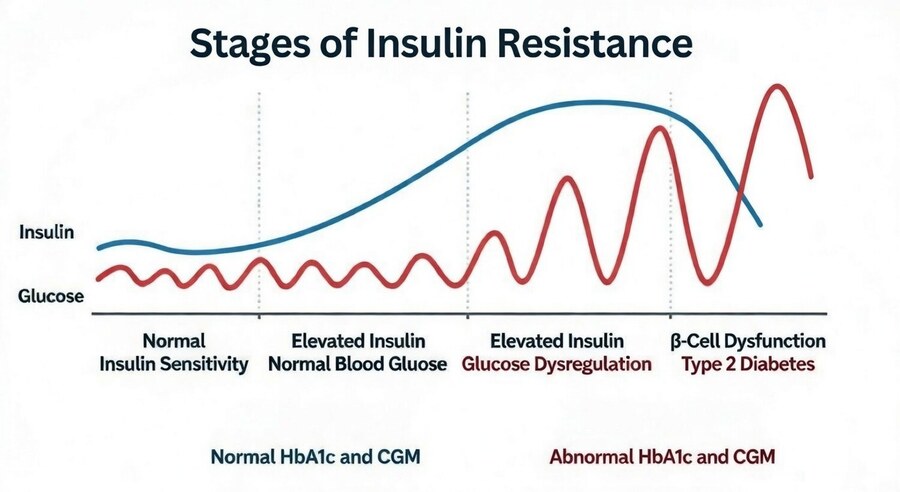
A few quick clarifications matter here, because this topic gets distorted fast. Insulin resistance is not the same thing as diabetes, and it doesn’t mean insulin is “bad”—you can’t live without it.
The issue is the growing requirement for higher and higher levels of insulin to get normal regulation. It’s also not strictly a “carb problem,” even though carbs can expose insulin resistance more obviously because they raise blood sugar more directly.
Insulin resistance is broader than that: it’s about how well your body handles fuel overall, including where energy ends up being stored and how efficiently tissues respond to the signals that manage it.
It is also not always something you can spot from a single lab value, because two people can have the same fasting glucose while one is maintaining it with far more insulin than the other. But there are many ways to spot it, as we’ll see later.
Insulin Resistance vs Prediabetes vs Type 2 Diabetes
| Stage | What’s happening | What glucose often looks like | What insulin often looks like |
| Early insulin resistance | Tissues less responsive; pancreas compensates | Normal | Higher than it should be |
| Prediabetes | Compensation strained; control less stable | Slightly elevated (fasting/A1C/OGTT) | Often high (can vary) |
| Type 2 diabetes | Compensation failing | Elevated | May be high early; can drop later as pancreas fatigues |
What Insulin Does
To understand insulin resistance, you first have to understand insulin’s actual job, because insulin isn’t a “fat-storage hormone”, or a villain that randomly ruins metabolism, and is largely misunderstood.
Insulin is closer to a fuel manager—and one of the main signals that switches your metabolism between the fed and fasted state. When you eat, especially when glucose enters the bloodstream, insulin rises to keep that fuel controlled and usable. In this “fed” mode, insulin helps move glucose into muscle cells, tells the liver to dial back its own glucose release because there’s already plenty circulating, and encourages the body to use what’s coming in and store any excess safely. When insulin falls between meals, the body shifts the other way, relying more on stored reserves to cover the gap.
The key message is simple: when fuel is coming in, the body prioritizes using and storing it, not pulling aggressively from reserves at the same time. That’s why fat release from fat tissue tends to dial down after meals and fat burning becomes less necessary in that moment.
People hear this and assume the signal is “the problem,” but it’s really just metabolic coordination—like not trying to unload and reload a warehouse at the same time. In a healthy system, this is temporary: the signal rises to process the meal, then falls again once the job is done.
That’s why blaming insulin misses the real issue. Insulin is responding to the environment you’re putting the system in—how much fuel is coming in, how often it’s coming in, and how much is being used. In most people, insulin is doing exactly what it’s designed to do: protect you from uncontrolled glucose and keep energy available.
The problem isn’t that insulin exists or that it spikes after meals; the problem is the chronic pattern where insulin needs to be high all the time just to maintain “normal.” And by the time someone reaches type 2 diabetes, it’s often not because insulin is too powerful—it’s because the pancreas can no longer keep up with the demand.
For a more in-depth look at exactly how insulin influences metabolic health, see this article here.
How Insulin Resistance Develops
Chances are, this didn’t start with one or two bad meals. Insulin resistance is what tends to happen after weeks, months, and sometimes years of running the system in the same direction: a fairly constant influx of energy, without enough demand to burn through it.
In that setting, the issue isn’t that insulin changes or “fails” — it’s that muscle and liver gradually become less responsive to insulin’s signal, so the pancreas has to turn the signal up (more insulin) just to keep blood sugar stable.
Importantly, you can see early shifts faster than most people realize: severe short-term overfeeding has been shown to measurably reduce insulin sensitivity in as little as 3 days in controlled conditions, while other studies show clear changes after around 2 weeks of overeating.
Meanwhile, reducing energy demand can do it too: one week of bed rest can significantly lower whole-body insulin sensitivity, and even one week of short sleep has been shown to reduce insulin sensitivity by nearly 30 percent. Over the long run, that’s why insulin resistance can build quietly — compensation works for a long time, until it doesn’t.
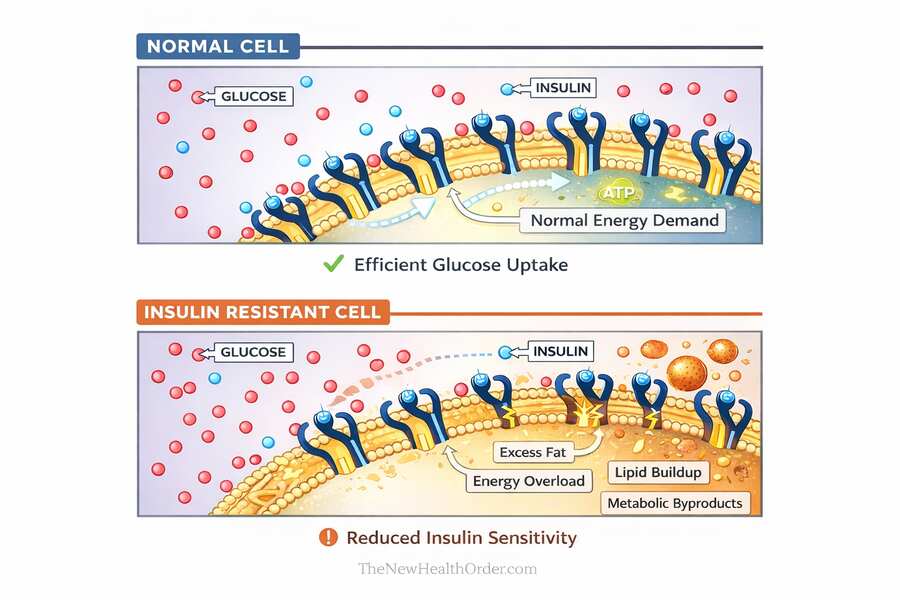
At the cellular level, the key shift isn’t that insulin “forces” glucose into cells, but that insulin sends a signal and tissues either respond strongly or weakly. In muscle and fat cells, a normal insulin signal moves glucose transporters to the cell surface so glucose can enter.
With chronic surplus, more energy starts spilling into places it wasn’t meant to be stored in large amounts—especially liver and muscle—and that internal fat creates metabolic “noise” that jams the signaling pathway. The message still arrives, but it doesn’t transmit cleanly, so the same insulin level produces less glucose uptake in muscle and less suppression of glucose output from the liver. That’s what reduced sensitivity actually means: you need more insulin to get the same result.
Here’s where the process becomes a loop, and this is the part most people miss. The pancreas isn’t listening to your cells and deciding to punish them with more insulin. It can’t see intracellular fat, signaling interference, or a muscle cell that’s already energy-replete.
What it can see is the bloodstream—especially rising or slow-clearing glucose after meals. And its main tool for dealing with that is insulin. So when glucose isn’t clearing efficiently, the pancreas turns up the volume and releases more insulin to keep blood sugar stable. For a long time that works. Glucose can stay normal precisely because insulin is working overtime in the background.
But overtime has a cost. If fat storage becomes less clean and more fatty acids spill into circulation, the liver and muscle get loaded even more, the signaling jam worsens, and the insulin requirement rises again. That’s the compensation cycle: weaker tissue response → higher insulin output → temporary control → more spillover → even weaker response.
Eventually some people hit a ceiling where the pancreas can’t keep increasing output, and that’s when blood sugar starts rising more obviously and the condition gets labeled as prediabetes or type 2 diabetes.
What Actually Causes Insulin Resistance?
For most people, the diagnosis comes later in the story. Long before blood sugar looks obviously abnormal, glucose can still appear “normal” on paper—but only because the body is holding it there with chronically higher insulin. That’s the compensation phase: insulin working overtime to keep fuel in the bloodstream under control. The real question, then, isn’t why insulin is high, but what’s creating the underlying fuel mismatch that keeps demanding more and more insulin in the first place.

Constant Fuel Availability
The simplest way to understand insulin resistance is in terms of energy in versus energy out. Your body handles energy best when intake is roughly matched to demand. You eat, insulin rises, fuel gets moved into cells or stored, and then insulin drops back down again. That rise-and-fall rhythm is normal.
The problem starts when energy is coming in more often than your body has a chance to use it. That can happen with ultra-processed foods, but it can also happen with genuinely healthy foods if portions and frequency consistently exceed demand.
Whole foods usually make this harder—because they’re more satiating and less calorie-dense—but the underlying biology is the same. When intake repeatedly outpaces output, the body spends more time in “fed mode,” insulin has to stay active more often to manage incoming fuel, and the system gradually becomes easier to overload.
In that environment, insulin isn’t doing anything wrong. It’s responding to the job it’s been given: keep the bloodstream stable by clearing incoming fuel. But when the system is constantly being fed, there’s rarely a true reset. Tissues spend more of the day in storage mode, less of the day drawing down what’s already stored, and the body gradually has to rely on higher insulin levels to maintain the same control.
Over time, that repeated pattern makes it easier for insulin sensitivity to drift downward, not because insulin is harmful, but because the whole system is being run at high throughput with few breaks.
A good metaphor here is a warehouse. If delivery trucks keep arriving faster than the warehouse can ship goods out, the staff doesn’t immediately give up—they start stacking boxes wherever there’s space just to keep the loading dock clear.
At first it works, but the warehouse slowly becomes cluttered, pathways get blocked, and moving goods becomes harder and slower. Constant fuel availability creates the same kind of problem metabolically: when energy keeps arriving faster than it’s being used, the body has to keep storing it to keep the bloodstream “clear,” and storage becomes less clean over time. That’s when insulin needs to rise higher to get the same result—and that’s the beginning of the insulin resistance loop.
This doesn’t mean you need extreme fasting or obsessive meal timing. It just means the body benefits from genuine gaps—periods where fuel isn’t constantly arriving—especially if your day-to-day movement isn’t high. When you restore that basic inflow/outflow rhythm, insulin gets to fall, tissues get a chance to “make room,” and the system becomes easier to regulate again.
Low Movement
If constant fuel availability is the “too much coming in” side of the equation, low movement is the “not enough going out” side. And this matters more than most people realize because movement isn’t just a way to burn calories; it’s one of the main reasons you have a large, active place to put incoming fuel in the first place. Your muscles are a massive energy sink. When you use them regularly, they pull glucose out of the bloodstream, refill and empty glycogen, and stay metabolically ready to accept more fuel the next time you eat. When you don’t, that capacity shrinks.
This is where modern life quietly breaks the system. If most of your day is sitting, your muscles are essentially closed off for hours at a time. Their fuel demand drops, glucose uptake drops, and you spend more time with tissues that are already relatively topped up.
So when food arrives, insulin has to work harder to clear the same amount of energy because the biggest consumer of that energy—active muscle—isn’t asking for much. In other words, low movement doesn’t just fail to compensate for overeating; it actively changes how easily your body can handle the same intake. That’s why athletes and those who are physically active can eat more calories without gaining fat.
There’s also a more direct effect that’s easy to miss: muscle contraction can pull glucose into muscle through signaling pathways that are at least partly independent of insulin. That’s why a simple post-meal walk can measurably blunt the spike—one randomized crossover study found a ~6% reduction in 2-hour glucose exposure (and about a ~10% lower peak) with a 10-minute easy walk immediately after a glucose load versus sitting.
And over the longer term, resistance training shifts baseline sensitivity too: in a 12-week randomized trial in women with metabolic syndrome, the resistance-training group reduced HOMA-IR by ~10% (4.13 → 3.72). In other words, you’re not “fixing metabolism” through willpower—you’re creating demand, making room, and restoring the normal flow of fuel through the system.
If we go back to the warehouse metaphor, movement is the shipping department. You can have a perfectly organized loading dock, but if almost nothing is being shipped out, the warehouse will eventually clog up no matter how efficient deliveries are. Low movement means the outflow is underpowered. And once outflow is underpowered, even a normal-looking inflow can start causing backlog, overflow into the wrong places, and rising insulin requirements.
This is why structured workouts help, but why they can’t fully rescue a day that is otherwise sedentary. The real goal isn’t just “exercise more.” It’s to build a day where your muscles are regularly turned on—often enough that fuel has somewhere to go.
Ectopic Fat (Fat in the Wrong Places)
Your body is designed to store large amounts of energy in fat tissue – that’s normal and protective. But when the inflow stays high and the outflow stays low for long enough, storage stops being perfectly “contained.” Like a bucket filling up with water beyond its capacity, more energy begins to spill into places that were never meant to act as long-term storage depots—especially the liver and skeletal muscle. That’s ectopic fat: fat stored in the wrong places.
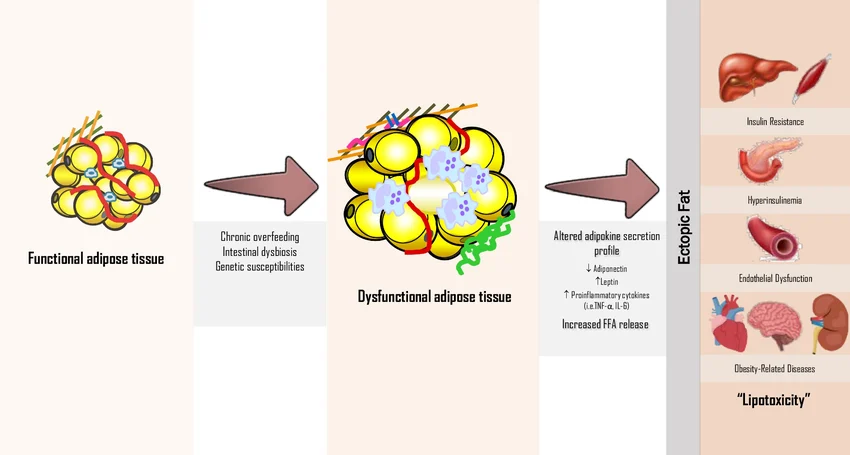
It’s worth being clear about cause and effect here. The buildup usually starts with the governing basics: energy surplus and low movement (i.e. energy in being consistently more than energy out).
But once fat starts accumulating in the liver and muscle, it doesn’t just sit there as a passive result. It becomes its own problem, because it actively makes fuel handling harder. Fat inside the liver and within muscle creates biochemical “static” that blunts insulin signaling.
In muscle, insulin normally helps move glucose transporters to the cell surface so glucose can enter efficiently. In the liver, insulin normally signals “we have fuel coming in, stop releasing extra glucose.” Ectopic fat interferes with both, so the same insulin level produces a weaker response. That’s the real meaning of reduced insulin sensitivity: the message still arrives, but the wiring doesn’t transmit it as cleanly.
This is also why insulin resistance can progress quietly. The pancreas compensates by producing more insulin to push the signal harder. For a while, that keeps glucose controlled and labs can look deceptively fine. But the underlying bottleneck remains, so insulin has to run higher more often, and the system becomes less stable over time.
Back to the warehouse: fat tissue is the proper storage area with shelves built for long-term inventory. Ectopic fat is what happens when the warehouse gets so backed up that boxes start getting stacked in the hallways and in front of doorways. You’re still “storing” things, but you’re storing them in a way that makes the whole operation slower and more chaotic. That’s what fat in the liver and muscle does metabolically: it turns what should be smooth fuel handling into a clogged, inefficient process.
Poor Sleep + Chronic Stress
Sleep and stress don’t usually create insulin resistance out of nowhere, but they can absolutely push the system in that direction. The reason is simple: when you sleep poorly or live in a constant state of pressure, your body behaves as if it needs to keep more fuel available just in case. Stress hormones make you a little more glucose-ready, a little more conservative with energy, and a little less interested in smooth, relaxed storage and clearance. This isn’t necessarily a problem with short-lived acute stressors, but chronic stress over months or years takes its toll on the metabolic system.
On the practical side, poor sleep and chronic stress also sabotage the inflow/outflow balance. You tend to be hungrier, cravings get louder, and it becomes easier to overeat without realizing it—especially later in the day. At the same time, you’re less likely to move, train, or choose the habits that keep insulin sensitivity high.
There’s also a more direct metabolic effect that matters. When stress is high, the body tends to increase liver glucose output, meaning the pancreas may need to produce more insulin to keep blood sugar stable, especially after meals. Again, insulin isn’t misbehaving; it’s responding to an environment that keeps telling the body that fuel availability must stay high.
The simplest takeaway is that sleep and stress shape the background conditions your metabolism is operating under. If you’re trying to improve insulin sensitivity while sleeping five hours, running on caffeine, and living in constant tension, you’re fighting with the thermostat turned the wrong way. Fixing sleep won’t magically override a chronic surplus, but it lowers the baseline strain so the core levers—food rhythm and movement—start working the way they’re supposed to.
Genetics
Genetics matters, but not in the fatalistic way people assume. It usually doesn’t “cause” insulin resistance on its own—it changes how much stress the system can tolerate before things start to back up. In other words, genetics mostly sets your threshold. Two people can live in the same modern environment, eat similarly, move similarly, and one will develop insulin resistance earlier while the other seems to get away with it longer. That difference is often genetics showing up as a smaller margin for error.
A big part of that margin comes down to how your body handles storage. Some people can expand fat tissue more easily and store surplus energy in a relatively safe way for longer. Others spill into the wrong places sooner (the liver and muscle) so the insulin resistance loop starts earlier even at a similar body weight.
Genetics also influences appetite regulation, how strongly you respond to sleep loss and stress, and how your liver manages glucose production. None of that guarantees an outcome, but it changes the slope of the hill you’re climbing.
Do not get caught using genetics as an excuse for your metabolic health – make it motivating rather than discouraging If your threshold is lower, it doesn’t mean you’re broken—it means the basics matter more.
The same levers still apply: Matching intake to output, building daily movement into your life, protecting sleep, and reducing chronic surplus are what keep the system stable. Genetics just determines how quickly you pay the price when those conditions drift in the wrong direction.
Causes of Insulin Resistance
| Driver | What it does | What you’ll notice | Best first lever |
| Constant fuel availability | Keeps insulin “on duty” | Hunger returns fast, grazing pattern | Clear meal spacing |
| Low movement | Shrinks outflow capacity | Energy swings, easier fat gain | Walk daily + post-meal walks |
| Ectopic fat | Jams insulin signaling | Fatty liver tendency, worsening labs | Reduce overload + train |
| Poor sleep/stress | Raises glucose output + appetite | Cravings, stubborn fasting glucose | Sleep hygiene + stress load |
| Genetics | Lowers threshold | Earlier onset at same lifestyle | Basics matter more, sooner |
Symptoms and Signs of Insulin Resistance
Insulin resistance can be sneaky because blood sugar can look “normal” for a long time while insulin is doing extra work in the background. So treat these as common patterns, not a self-diagnosis.
Common Everyday Signs of Insulin Resistance
- Post-meal crashes: sleepy, foggy, irritable, or suddenly hungry again 1–3 hours after eating (especially after carb-heavy meals).
- Excessive hunger: you get hungry quickly after eating, feel snacky, or struggle to feel satisfied.
- Cravings: strong pull toward sweets, refined carbs, or late-night eating.
- Energy that swings: you feel good for a bit, then dip hard; you may feel dependent on caffeine or frequent meals.
- Poor tolerance to gaps between meals: you feel shaky, edgy, or overly distracted by food when you go longer without eating (often a metabolic flexibility issue).
- Weight gain: especially around the midsection over time (waist size creeping up).
- Harder fat loss even when effort is consistent (often because hunger and regulation are fighting you).
- Fatty liver tendency (sometimes found on imaging or suggested by labs).
- Skin tags (more common in insulin-resistant states, though not exclusive).
- Darkened, velvety skin in folds (neck/armpits/groin; acanthosis nigricans), a stronger clue when present.
Lab-Pattern Clues (Often more Reliable than Symptoms)
- Higher fasting insulin (early marker, often before glucose changes).
- Higher triglycerides and lower HDL (a common metabolic pattern).
- Rising fasting glucose or A1C over time (often later-stage signals).
- Blood pressure trending up (part of the broader insulin-resistance cluster).
If you recognize several of these at once, the best move isn’t to guess but confirming with the right measurements (fasting insulin and related markers) and then tackling the drivers you already understand: reduce chronic fuel overload, increase daily movement, and protect sleep.
How To Measure Insulin Resistance
If insulin resistance requires needing more insulin to get the same job done, then the best measurements are the ones that look at insulin itself, not just glucose. The problem is that most routine checkups don’t include insulin, so people get told everything is fine while the compensation phase is already underway. Think of measurement in layers: the more directly you measure insulin and its effects, the earlier you can see the issue.
1. Fasting glucose
Fasting glucose is the amount of glucose in your blood after an overnight fast (typically 8–12 hours). It’s usually reported in mg/dL in the U.S. (or mmol/L elsewhere) and is one of the most common screening tests for prediabetes and diabetes because it’s simple and widely available.
You can measure fasting glucose either with a lab blood draw (most consistent) or at home with a fingerstick glucose meter (more convenient, but less precise and sensitive to technique). A CGM can also show your fasting levels and overnight trends, though it still benefits from occasional lab confirmation.
It’s useful, but it has a limitation: fasting glucose can look normal for a long time even while insulin is already elevated and doing extra work. It’s also influenced by sleep, stress, illness, and hard training because fasting glucose is heavily driven by liver glucose output overnight. That’s why fasting glucose is best treated as one dashboard gauge—most informative when paired with fasting insulin (or HOMA-IR) and tracked over time rather than taken as a single definitive answer.
It’s also cheap and simple to monitor. My wife and I measure our glucose and ketone levels every Saturday morning with a simple glucose monitor (we use this Keto-mojo branded finger stick device). Over time, it allows you to build a profile.
2. Fasting Insulin
This is the cleanest place to start and it’s also one of the best early signals. If fasting insulin (the amount of insulin circulating in your blood after an overnight fast) is elevated, it often means your body is having to run a stronger baseline insulin signal just to keep fasting glucose stable. It’s not perfect on its own (it’s a snapshot of one brief moment in time), but it’s one of the most direct early indicators that you’re compensating.
3. HOMA-IR (fasting insulin + fasting glucose)
HOMA-IR is a simple calculated estimate of insulin resistance using two fasting measurements taken at the same time: fasting glucose and fasting insulin. You don’t measure HOMA-IR directly—you measure those two numbers after an overnight fast, then use a standard formula to combine them into a single score.
The benefit is that it captures the relationship between glucose and insulin. Glucose can look “normal” for a long time because the pancreas is compensating by producing more insulin. HOMA-IR helps reveal that early pattern by showing when glucose is being held in range only because insulin is running higher than it should.
In terms of practicality, glucose can be measured at home, but insulin usually requires a lab test (or an at-home kit that still gets processed by a lab).
4. Oral Glucose Tolerance Testing (OGTT)
An OGTT is a way to see how your body handles a controlled glucose challenge. After an overnight fast, you drink a standardized glucose solution and then have blood drawn at set times (commonly at baseline and 2 hours, sometimes with additional points like 30 or 60 minutes) to track how quickly glucose returns toward normal.
A standard OGTT measures glucose only, which is helpful for diagnosing impaired glucose tolerance or diabetes. But the more informative version also measures insulin at the same time points. That shows whether your body is keeping glucose “normal” only by producing a disproportionately large insulin response—basically insulin resistance caught in real time.
5. A1C
A1C (hemoglobin A1c) is a blood test that estimates your average blood sugar over the past 2–3 months by measuring how much glucose has attached to hemoglobin in your red blood cells. It’s typically done as a lab test from a standard blood draw, although some clinics can run a quick point-of-care version from a fingerstick.
It’s useful because it smooths out day-to-day ups and downs and gives a longer-term trend, which is why it’s commonly used to flag prediabetes and type 2 diabetes and to track progress over time. The limitation is that it can miss early insulin resistance: glucose can stay “normal” for a long time if your pancreas is compensating by producing extra insulin. So A1C is a solid big-picture marker of glucose control, but it’s not the earliest warning light for insulin resistance.
6. Helpful “proxy” patterns
These aren’t direct measures of insulin resistance, but they often correlate with it and can strengthen the picture:
- Triglycerides and HDL (a common pattern is higher triglycerides and lower HDL)
- Waist circumference / visceral fat trend
- Blood pressure trend
- Fatty liver indicators (imaging or liver enzymes, depending on context)
How to Improve Insulin Sensitivity
Once you understand insulin resistance as a fuel-handling backlog, the solution becomes much less mysterious. You simply have to begin rebuilding the conditions where insulin doesn’t have to shout all day. The most reliable improvements come from a few boring fundamentals that consistently reduce fuel overload, increase demand, and clear spillover from the wrong places.
- End the chronic mismatch (fuel in vs fuel out). Insulin sensitivity improves when intake stops consistently outpacing energy use. You don’t need perfect calorie tracking, but you do need the ongoing surplus to stop.
- Restore meal spacing so insulin can fall. Cut the constant drip-feed of snacks, grazing, and liquid calories. Clear gaps between meals give the system time to run on stored fuel and “make room” again.
- Walk after meals. This is one of the highest-ROI habits because it directly increases glucose disposal when it matters most. Even a short, easy post-meal walk has been shown to reduce post-meal glucose exposure by about ~6% (and peak glucose by roughly ~10%) compared with sitting.
- Lift weights consistently. Resistance training increases metabolic capacity (muscle is a major sink for glucose) and improves insulin-resistance markers over time. In a 12-week randomized trial in women with metabolic syndrome, resistance training reduced HOMA-IR by ~10%.
- Break up long sitting. A few workouts a week help, but they don’t fully cancel a day spent in “low-demand mode.” Frequent low-intensity movement keeps muscles metabolically active and reduces backlog.
- Protect sleep and lower chronic stress. These aren’t “soft” factors; they directly affect hunger, cravings, recovery, and liver glucose output. Improving them makes the core levers above dramatically easier to sustain.
- Temporarily reduce carbs to lower the insulin load. If you’re already trending insulin resistant, lowering carbohydrate intake is one of the most direct ways to reduce the glucose load you’re asking the system to handle, which usually means a smaller insulin response is required to keep blood sugar controlled. That’s not controversial—it’s basic physiology. You don’t necessarily have to do it forever, but as a short-to-medium-term strategy it can be a powerful way to stop insulin running overtime while you rebuild the fundamentals (movement, meal spacing, sleep, and overall energy balance). Once things stabilize, many people can reintroduce carbs in a way that matches their activity and tolerance rather than going straight back to constant high-load intake.
See this article here for a deeper dive in low-carbohydrate and Ketogenic diets.
Final Thoughts
Insulin resistance is easy to misunderstand because it often starts invisibly. In the early stages, blood sugar can look normal and life can feel mostly the same, but only because the body is compensating—producing more insulin to maintain control. The problem isn’t insulin “going bad.” The problem is that muscle and liver stop responding as well to insulin’s signal, usually after a long stretch of fuel coming in more reliably than it’s being used. Once that happens, the pancreas has one main tool to keep the bloodstream stable: turn the signal up.
The encouraging part is that insulin resistance is also one of the most fixable metabolic problems when you tackle it at the right level. You don’t need magic supplements or complicated protocols. You need to restore flow: reduce the chronic backlog of incoming energy, increase daily movement so fuel has somewhere to go, and give the system enough space for insulin to fall between meals. If you’re already showing signs of resistance, lowering carbohydrate intake can be a particularly powerful lever—not forever, but long enough to reduce glucose load and stop insulin running overtime while the deeper drivers improve.
Most importantly, don’t get distracted by blaming insulin, carbs, or any single food. Insulin is doing what it’s designed to do: protect you from uncontrolled glucose. The real win is building a lifestyle where it doesn’t have to shout all day to do that job.
FAQs
What is insulin resistance, in simple terms?
Insulin resistance means your body needs more insulin than it should to keep blood sugar and fuel under control. Insulin is the signal; the issue is that muscle and liver become less responsive to it. Early on, blood sugar can look normal because the pancreas compensates by producing extra insulin. Over time, that “overtime” becomes harder to sustain, and glucose regulation can start to slip.
Can you have insulin resistance with normal blood sugar?
Yes. This is common early on because the pancreas can increase insulin output and keep glucose in range for years. That’s why fasting glucose alone can miss early insulin resistance. Tests like fasting insulin and HOMA-IR help reveal whether glucose is being held “normal” only because insulin is running high in the background.
What’s the fastest way to improve insulin sensitivity?
The fastest improvements usually come from lowering the daily fuel load and increasing demand. Start with clear meal spacing (stop constant grazing), add walking—especially after meals—and do resistance training consistently. If you’re already insulin resistant, temporarily reducing carbs can reduce glucose load and the insulin required to manage it, making it easier for the system to recover while you rebuild the fundamentals.