Omega-3 and omega-6 are two of the most talked-about fats in nutrition, yet they are often explained in the most simplistic way possible. Omega-3 is praised, omega-6 is blamed, and somewhere in the middle people are told to worry about a ratio few can clearly define. The reality is more nuanced — and more interesting.
These fats are not just calories. They are built into the body’s cells and help shape everything from inflammation to brain function to cardiovascular health. In this article, we will look at what omega-3 and omega-6 fats actually are, what they do in the body, why the ratio between them became such a talking point, and what the science really says about whether that balance matters.
What Are Omega-3 and Omega-6 Fats?
Omega-3 and omega-6 fats are both polyunsaturated fats (PUFAs), which simply means they have more than one double bond in their structure. The “omega-3” or “omega-6” label simply tells you where the first double bond sits when counted from the methyl end of the fatty acid chain. While the technicals can get complicated, the important takeaway is simpler: these are two different families of fat, and the body uses them differently. Both matter, and both are normal parts of human biology.
See this article here for an in-depth look at the different types of fats and how they affect the body.
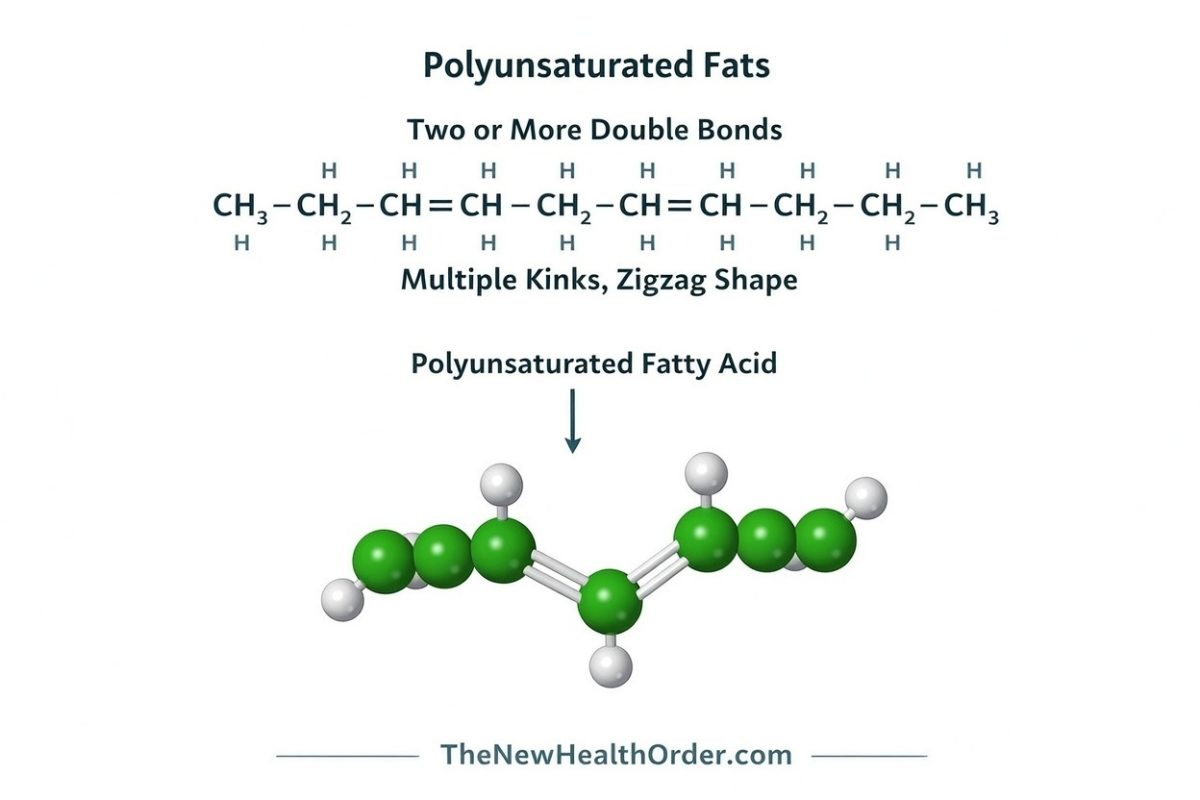
They are also both considered essential fats — fats the body must obtain from food because it cannot produce them in sufficient amounts on its own. Humans can only insert double bonds up to a certain point in a fatty acid chain, which means we cannot make the parent omega-3 fat alpha-linolenic acid (ALA) or the parent omega-6 fat linoleic acid (LA) from scratch. Those two have to come from food. From there, the body can do some conversion work, but only within limits.
One important clarification up front: when people talk about omega-3 or omega-6, they often make it sound as though each category is just one thing, which it is not. Each family contains several different fats, and they do not all behave the same way, important because a conversation about flaxseed is not quite the same as a conversation about salmon, and a conversation about walnuts is not quite the same as a conversation about arachidonic acid in cell membranes.

The Main Omega-3 Fats
The three omega-3 fats that matter most in nutrition are alpha-linolenic acid (ALA), eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA). ALA is the shorter-chain omega-3 found mainly in plant foods such as flax, chia, walnuts, canola, and soy, whereas EPA and DHA are the longer-chain omega-3s found mainly in seafood and algae.
| Fat | Full name | Found mainly in | Can the body make it? | Key point |
| ALA | Alpha-linolenic acid | Flax, chia, walnuts, canola, soy | No — must come from food | Essential plant omega-3; conversion to EPA/DHA is limited |
| EPA | Eicosapentaenoic acid | Fatty fish, seafood, algae | Only limited amounts from ALA | Important in signaling pathways and cardiovascular health |
| DHA | Docosahexaenoic acid | Fatty fish, seafood, algae | Only very limited amounts from ALA | Especially important in the brain and retina |
In NIH food tables, just one tablespoon of flaxseed oil provides about 7 g of ALA, one ounce of chia seeds provides about 5.06 g, while a 3-ounce serving of farmed Atlantic salmon provides about 1.24 g of DHA and 0.59 g of EPA.
To put that in perspective, adult adequate intake for ALA is around 1.1 g per day for women and 1.6 g per day for men, while EPA and DHA do not have a formal daily recommended intake, with most guidance instead encouraging regular fish consumption. That means foods like flaxseed oil and chia seeds can easily exceed daily ALA needs, while a single 3-ounce serving of salmon can provide a substantial amount of preformed DHA and EPA.
That difference between ALA and EPA/DHA is important. The body can convert some ALA into EPA and then into DHA, but the conversion is limited—the NIH fact sheet notes reported conversion rates of less than 15 percent—and reviews generally find that raising ALA intake tends to increase EPA more reliably than DHA.In other words, plant omega-3s (ALA) are real omega-3s, but they are not interchangeable with the EPA and DHA found directly in marine foods.
DHA in particular is extremely important, since it is heavily concentrated in tissues where membrane structure matters. Reviews note that DHA is the quantitatively most important omega-3 in the brain, and in the retina it is even more striking: DHA makes up roughly 50–60 percent of the total fatty acids in rod outer segments of photoreceptors. That helps explain why omega-3s are often discussed in relation to brain and eye function rather than just heart health.

The Main Omega-6 Fats
On the omega-6 side, the two most important fats to understand are linoleic acid (LA) and arachidonic acid (AA). LA is the essential parent omega-6 fat (the main omega-6 fat the body gets from food and uses as a starting point to make other omega-6s). It is widely found in nuts, seeds, and many plant oils, especially soybean, corn, sunflower, and grapeseed oils.
AA is a longer-chain omega-6 fat found in animal foods and also produced in the body from LA. So just as ALA sits upstream of EPA and DHA in the omega-3 family, LA sits upstream of AA in the omega-6 family.
| Fat | Full name | Found mainly in | Can the body make it? | Key point |
|---|---|---|---|---|
| LA | Linoleic acid | Seeds, nuts, soybean oil, corn oil, sunflower oil | No — must come from food | Essential parent omega-6 fat |
| AA | Arachidonic acid | Meat, poultry, eggs; also made from LA | Yes, from LA | Involved in signaling compounds tied to inflammation, clotting, and vascular responses |
Omega-6s is often where the topic often gets oversimplified, however. Omega-6 is commonly portrayed as the “bad” side of the omega story, but LA is still an essential nutrient the body needs and cannot make itself. It is built into membrane phospholipids, contributes to skin ceramides, and helps maintain the epidermal water barrier.
True omega-6 deficiency is rare in otherwise healthy adults, largely because linoleic acid is now so widespread in the food supply. So the real concern is usually not the presence of omega-6 itself, but the sheer amount consumed in modern diets — especially from processed foods and seed oils — often alongside relatively low omega-3 intake.
For a look into which seed oils and what the science says about their effect on metabolic health, see this article here.

What do Omega-6 and Omega-3 do in the Body?
Once in your system, omega fatty acids don’t just act as calories—they become part of your cell membranes and also serve as raw materials for signaling molecules that influence how your body responds to stress, injury, and immune triggers.
A big part of their role is that they help shape the body’s inflammatory signaling environment. Not inflammation in the vague “bad thing” sense, but inflammation as a normal biological tool: the body’s way of initiating a response to damage or threat and then repairing and returning to baseline afterward. These fats can be converted into messenger molecules that tell tissues when to ramp a response up and when to cool it down.
In broad terms, omega-6 pathways tend to be more involved in initiating inflammatory responses—helping the body mobilize, clot, and react when something happens. Omega-3 pathways tend to be more involved in moderating and resolving inflammation—supporting the mechanisms that calm the response and wind down inflammatory signals once the job is done.
The important point is that both roles are useful in the right amounts. And when either a below the necessary levels, bad health effects will begin to show. The goal isn’t to eliminate inflammation; it’s to have the right response at the right time, followed by proper resolution. That’s why the modern conversation usually isn’t “omega-6 vs omega-3,” but whether the overall balance in the diet supports that full cycle.
Why Is Omega-6 Often Called Inflammatory?
Omega-6 gets that reputation mainly because one of its downstream fats, arachidonic acid (AA), can be used to make compounds such as prostaglandins, thromboxanes, and leukotrienes, many of which are involved in inflammation, clotting, and vascular responses.
More broadly, the NIH notes that eicosanoids made from omega-6 fats are generally more potent mediators of inflammation, vasoconstriction, and platelet aggregation than those made from omega-3 fats, though there are important exceptions.
What many fail to appreciate, however, is that inflammation is not automatically harmful. In fact it is a normal part of physiology and is vital for immune defense and tissue repair.
Some assume that eating more omega-6 must inevitably increase inflammation, as though there is a threshold beyond which a normal physiological process becomes excessive and harmful. The logic is understandable, but the evidence is less straightforward: studies suggest that increasing linoleic acid intake — the main dietary omega-6 fat and a precursor to arachidonic acid — does not automatically translate into a meaningful rise in inflammation in humans.
In a systematic review of randomized controlled trials in healthy people, for example, researchers found virtually no evidence that increasing dietary linoleic acid raised common inflammatory markers such as C-reactive protein, TNF-alpha, or inflammatory cytokines. So it is fair to say omega-6 can feed pathways involved in inflammation (for our benefit), but it is too simplistic to say omega-6 intake itself causes inflammation beyond what is normal.
Why Is Omega-3 Considered Beneficial?
Omega-3s, especially eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), are often viewed more positively because they tend to shift the body’s signaling environment in a less inflammatory direction. They become part of cell membranes and can compete with arachidonic acid in some of the same pathways, which changes the kinds of compounds the body produces.
Instead of pushing as strongly toward inflammatory signaling, omega-3s help favor compounds that are calmer and more balanced. They can also be converted into molecules such as resolvins, protectins, and maresins, which help bring inflammation to a close once it has served its purpose.
For example, in a meta-analysis of 38 randomized controlled trials involving 149,051 participants, omega-3 fatty acids were associated with lower cardiovascular mortality, fewer non-fatal heart attacks, and fewer major coronary events.
Another meta-analysis found that EPA and DHA lowered triglycerides in a near-linear way as intake increased. So the case for omega-3 is not just theoretical; it also shows up in real clinical outcomes, especially in cardiovascular health. That said, the evidence is not entirely one-sided, since benefits can vary by dose and form, and some analyses have linked higher-dose omega-3 supplements with a greater risk of atrial fibrillation.
So the simplest way to think about it is this: omega-6 and omega-3 are both biologically active, essential fats. The difference isn’t that one matters and the other doesn’t—it’s that they tend to shape the body’s signaling environment in different directions. Omega-6 pathways are often involved in starting inflammatory responses (which is sometimes exactly what you want), while omega-3 pathways are often involved in moderating and resolving inflammation. In the right balance, both are useful—because the goal isn’t “no inflammation,” it’s appropriate inflammation at the right time, followed by resolution.

Get Your Free Book!
Enter your email to get the free book and occasional updates from The New Health Order.
Does the Omega-6 to Omega-3 Ratio Matter?
Most are in alignment and agreement with everything we have said up to now. Omega-6 and omega-3 are both essential, both biologically active, and both shape the body’s signaling environment. Where the paths split and disagreements arise is when talking about whether there’s a limit to omega-6 intake—where normal, useful inflammatory signaling starts tipping into chronic, unhelpful signaling—and whether there’s an optimal omega-6 to omega-3 ratio we should aim for.
On the surface, the ratio sounds like a neat solution. You’ll often hear targets floated anywhere from roughly 1:1, to 2:1, to 4:1, and then you’ll see estimates that many modern Western diets sit far higher—often quoted around 10:1 to 20:1 (or even 20–25:1) for omega-6:omega-3. People also point to anthropological/evolutionary estimates suggesting historical patterns may have been closer to ~1:1.
Some studies have also reported blood fatty-acid ratios in high–marine food populations. For example, plasma phospholipid data in Inuit groups report omega-6:omega-3 ratios around ~1.5:1 (derived from n-3:n-6 = 0.67) in Greenland Inuit women, and ~3.2:1 (derived from n-3:n-6 = 0.31) in Nunavik Inuit. These biomarker ratios don’t prove a single “correct” dietary ratio, but they do illustrate how different the underlying fatty-acid balance can look across populations and dietary patterns.
The issue with using the ratio as a guide for health, however, is that it is a blunt tool. It compresses two variables into one number, and that number can change in multiple ways. A ratio can improve because omega-3 increases, because omega-6 decreases, or because both change—and those aren’t equivalent interventions. You can even create a “better” ratio on paper while still being low in omega-3, simply by cutting omega-6. That’s why the ratio is better treated as a signal than a target.
What Ratio Research Suggests
When researchers have looked at omega-6:omega-3 ratios (rather than omega-6 or omega-3 in isolation), the signal is mixed but not meaningless: lower ratios are often reported alongside better outcomes in certain disease contexts, while higher ratios tend to track with worse outcomes in some observational datasets.
A commonly cited review by Simopoulos summarizes multiple lines of ratio-based findings and also frames the historical contrast (roughly ~1:1 vs Western diets often quoted around ~15:1–16.7:1). In that review’s summary of prior clinical and epidemiologic studies, a 4:1 ratio is described as being associated with a large reduction in total mortality in secondary prevention of cardiovascular disease, 2.5:1 is described as reducing rectal cell proliferation in colorectal cancer patients (while 4:1 with similar omega-3 did not), and ratios around 2–3:1 (rheumatoid arthritis) and 5:1 (asthma) are described as beneficial, with 10:1 described as having adverse consequences in an asthma context.
More recently, large prospective biomarker work has examined ratios directly. In a UK Biobank analysis of 85,425 participants using plasma fatty-acid measures, people in the highest omega-6/omega-3 ratio quintile (median ratio ~14.8) had higher mortality rates than those in the lowest quintile (median ratio ~5.9): about 26% higher all-cause mortality (HR 1.26), 31% higher cardiovascular mortality (HR 1.31), and a smaller, weaker association for cancer mortality at about 14% higher (HR 1.14, borderline).
Taken together, the ratio literature supports a cautious conclusion: ratio differences can correlate with meaningful health outcomes, and “lower ratio” patterns sometimes show up in better clinical results. But the findings don’t converge cleanly on one universal “perfect ratio,” and different conditions are often associated with different ratio ranges in the literature.
Better ways to Assess Omega-6 and Omega-3 Balance
The omega-6:omega-3 ratio is a tempting shortcut as it would be a convenient and easy to measure metric—but it’s a weak measuring stick because it can move in opposite ways (you can lower the ratio by raising omega-3, by lowering omega-6, or by changing both), and those different paths don’t imply the same biology.
That’s why a UK Food Standards Agency workshop review we saw earlier, while measuring poorer outcomes with higher ratios, also concluded that the dietary n-6:n-3 ratio is not a useful concept for cardiovascular health and can distract from more meaningful markers—especially absolute long-chain omega-3 intake/status.
A more useful approach, therefore, may be to separate (1) what you eat from (2) what shows up in the body, and then ask what happens when each fatty-acid family is changed independently.
What Happens When you Increase Omega-3 Independently
We’ll start with omega-3 because this is where the evidence is strongest. In a large meta-analysis of randomized trials (38 RCTs; ~149,000 participants), omega-3 supplementation was linked to modest reductions in several cardiovascular outcomes, including cardiovascular mortality and non-fatal MI (non-fatal heart attacks).
The importance here is that it shows that raising omega-3—especially the long-chain forms EPA and DHA—can produce measurable changes in controlled trials. In other words, ensuring sufficient omega-3 status is a practical lever beyond just hitting a ratio.
In practice, that means “omega-3 adequacy” isn’t just a ratio issue; it’s an intake/status issue. EPA and DHA are the forms most consistently emphasized for cardiovascular support, and the American Heart Association recommends eating fish (particularly fatty fish) at least twice per week. They also explicitly note that algae supplements with EPA and DHA (typically 250-500 mg/day) are an option for people who do not consume fish or other animal products.
Otherwise Plant omega-3 (ALA) still matters, but the body converts only small amounts to EPA/DHA, so ALA alone may not reliably produce the same tissue EPA/DHA status.
What Happens When you Increase Omega-6 Independently
For the common claim that “more omega-6 drives systemic inflammation,” the best human evidence is narrower and less dramatic than many people assume. In controlled trials, where researchers increased linoleic acid (LA) specifically, the overall signal is mostly neutral: a systematic review of randomized controlled trials in healthy people found “virtually no evidence” that adding LA meaningfully increased a broad range of inflammatory markers such as CRP, cytokines, adhesion molecules, or TNF-α. A later meta-analysis of 30 randomized trials reached much the same conclusion, finding no significant overall effect of higher LA intake on common blood inflammatory markers.
For harder outcomes, the signal in prospective cohort meta-analyses is generally neutral-to-favorable rather than harmful. Higher dietary LA intake has been associated with lower risk of coronary heart disease events and CHD death, and higher dietary or biomarker LA has also been associated with modestly lower all-cause and cardiovascular mortality. These are observational findings, so they do not prove causality, but they do show that the human evidence does not neatly support the idea that more dietary LA simply translates into worse health.
At the same time, these studies should be interpreted carefully. Most of the controlled trials were designed to test LA itself under controlled dietary substitutions, not the kinds of omega-6 exposures common in modern life such as repeatedly heated frying oils, industrially processed foods, or oxidized lipid byproducts. That is an important distinction. The evidence is fairly reassuring when the question is “does more linoleic acid, by itself, reliably raise systemic inflammation in humans?” But it is much less definitive when the question becomes “what happens when people consume large amounts of omega-6-rich oils in heavily processed and repeatedly heated food environments?”
What Happens when you Reduce Omega-6 Independently
Here, the evidence is more biochemical than clinical. In other words, researchers can see certain blood markers change, but that is not the same as proving a reduction in disease. Some studies suggest that lowering linoleic acid reduces certain oxidized byproducts in the bloodstream, which supports the idea that omega-6 intake can affect these downstream compounds. Still, that alone does not prove that lowering omega-6 leads to better long-term health outcomes.
It also helps to keep one important detail in mind: the body does not appear to convert linoleic acid into arachidonic acid in a simple, unlimited way. In adults eating typical Western diets, even fairly large changes in linoleic acid intake do not seem to produce major changes in blood arachidonic acid levels. So while the common story is that “more omega-6 means much more arachidonic acid and therefore more inflammation,” the real picture appears to be more restrained than that.
What this Does and Does Not Indicate
What we have, right now, is stronger evidence for the benefits (and tradeoffs) of improving omega-3 status than we have for broad benefits of omega-6 restriction. We also have decent evidence that simply increasing linoleic acid does not reliably raise systemic inflammation markers in healthy people, and observational data that higher linoleic acid intake is not associated with worse CHD outcomes in aggregate.
What we do not have is a large set of long-duration modern trials that isolate “omega-6 lowering” (especially in the real-world form people worry about—ultra-processed, high-heat oil exposure) and then track hard cardiometabolic endpoints.
That’s why the most defensible assessment today is: treat omega-3 as a status problem you can measure and correct (dietary intake and/or Omega-3 Index), and treat omega-6 less as a single number and more as a source/context question—how much is coming from whole foods versus industrial, frequently heated, ultra-processed sources.
Final Thoughts
Omega-3 and omega-6 fats are not “good fat” and “bad fat” in any simplistic sense. Both are essential, both play real roles in the body, and both are involved in important signaling pathways. The problem is not that omega-6 exists at all. It is that modern diets often deliver it in large amounts—especially through processed foods and heated seed oils—while omega-3 intake tends to stay relatively low.
That is also why the omega-6 to omega-3 ratio should be treated carefully. It can be a useful shorthand for imbalance, and there is data that relate higher ratios to worsening cardiovascular outcomes, but it is not a magic number that explains everything by itself. A high ratio can reflect too little omega-3, too much omega-6, or both. In many cases, the more meaningful question is not simply what the ratio is, but what the diet actually looks like in practice.
So the takeaway is not that everyone should fear omega-6, nor that omega-3 is a miracle cure. It is that these fats shape the body in different ways, and the balance between them matters. Understanding that balance helps make sense of why modern dietary patterns may push the body toward a less favorable inflammatory environment—even if the science is more nuanced than the headlines suggest.
FAQs
What is the difference between omega-3 and omega-6 fats?
Omega-3 and omega-6 are both essential polyunsaturated fats, meaning the body needs them from food. Omega-3s are more associated with inflammation resolution and brain and eye health, while omega-6s are more involved in inflammatory signaling and normal immune response.
Is omega-6 bad for you?
Not inherently. Omega-6 is an essential fat the body needs and cannot make on its own. The bigger concern is that modern diets often provide large amounts of omega-6 from processed foods and heated seed oils while omega-3 intake stays relatively low.
Does the omega-6 to omega-3 ratio really matter?
It matters, but not as a perfect health target. A high ratio can reflect too little omega-3, too much omega-6, or both. That is why the ratio is better viewed as a clue about imbalance rather than a magic number on its own.